Agentic Orchestration Goes Pharma: How 310,000-Partner Networks Are Deploying Governed AI Agents Across Life Sciences Supply Chains

The pharmaceutical supply chain is one of the most complex, regulated, and high-stakes logistics networks on the planet. Now it's becoming one of the first to deploy governed AI agents at scale — and the implications for healthcare logistics are profound.
The 310,000-Partner Network Goes Agentic
TraceLink, the life sciences industry's largest digital supply chain network, announced in February 2026 that it is scaling agentic orchestration across its global healthcare supply chain platform. The numbers behind this shift are staggering:
- 310,000+ trading partners connected across the network
- 339,000+ active links between organizations
- 7 billion+ regulated transactions processed annually
- 971% year-over-year growth in MINT (Multienterprise Information Network Tower) live links
- 59% growth in MINT customers year over year
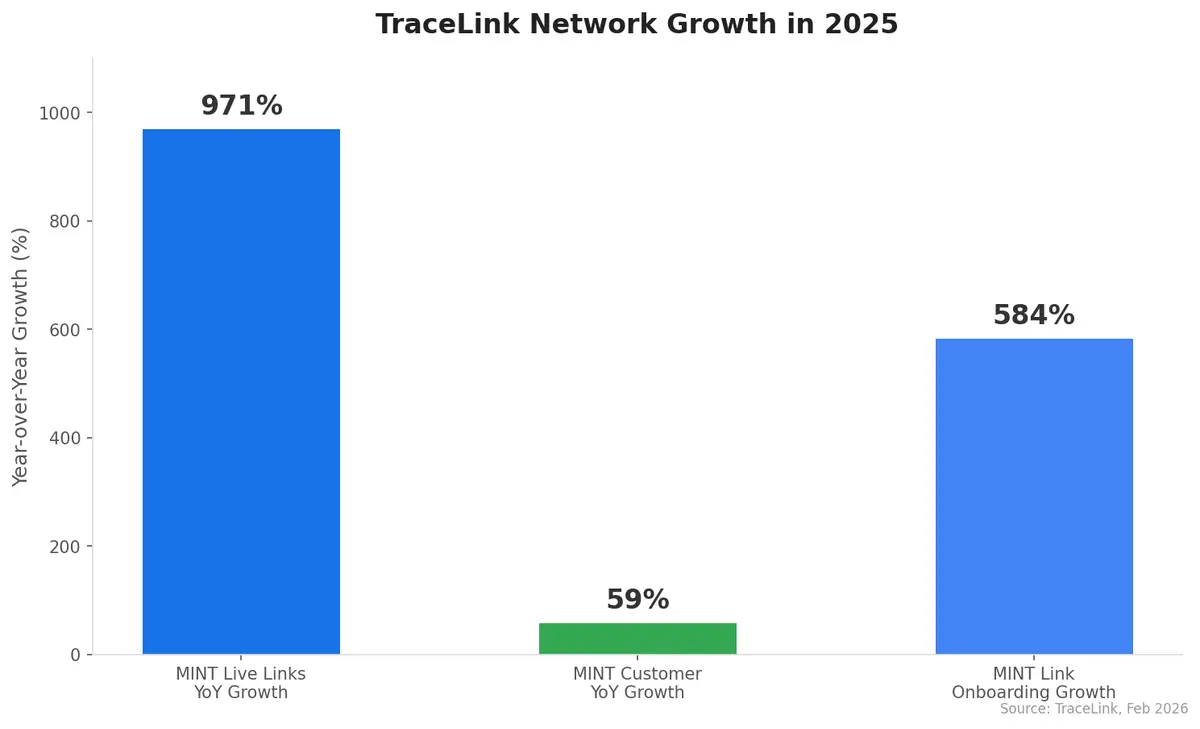
"Digitalization and the operational deployment of AI across one of the world's most valuable and least automated industries represents a generational opportunity," said Shabbir Dahod, TraceLink's President and CEO. The company's strategy centers on what it calls the "network-is-the-product" model — where every transaction processed and every partner connected strengthens shared digital infrastructure for all participants.
Why Pharma Is Different: Compliance Before Intelligence
Unlike general freight or retail logistics, pharmaceutical supply chains operate under layers of regulatory mandates that make AI deployment fundamentally different. You can't deploy autonomous agents in an environment where a single compliance failure can result in product recalls, patient harm, or nine-figure fines.
The U.S. Drug Supply Chain Security Act (DSCSA) — which reached full enforcement milestones in 2025 with manufacturer compliance by May and wholesaler compliance by August — requires every prescription drug to be serialized and tracked at the package level across the entire chain of custody. During peak DSCSA implementation, TraceLink was activating approximately 300 new live links per week and processing billions of serialized EPCIS transaction events.
This compliance infrastructure isn't just a regulatory checkbox. It's the foundation that makes agentic orchestration possible. Governed AI agents need structured, verified, auditable data to operate — and serialization mandates created exactly that.
From Compliance Backbone to Agentic Intelligence
The shift from compliance to orchestration is the key development. In 2025, TraceLink launched 21 new transaction workflows spanning transportation, manufacturing, logistics, and commercial operations — activating 16 transaction types across five core business orchestrations and running 182 live transactions in production.
What does agentic orchestration actually look like in pharma? Consider these cross-enterprise scenarios:
Automated Exception Handling: When a temperature excursion is detected in a cold chain shipment of biologics, an AI agent can simultaneously alert the manufacturer, reroute the shipment to the nearest compliant facility, trigger quality hold documentation, and notify downstream distributors — all within minutes, across multiple organizations, with full audit trails.
Serialization Verification at Scale: Instead of manual verification checks at each handoff point, governed agents continuously validate serialized data as products move from manufacturer to wholesaler to pharmacy, flagging discrepancies before they become compliance violations.
Predictive Demand Orchestration: AI agents analyzing prescription fill data across pharmacy networks can signal upstream manufacturers about demand shifts weeks before traditional forecasting methods, enabling proactive production and distribution adjustments.
The Pharma 4.0 Market Surge
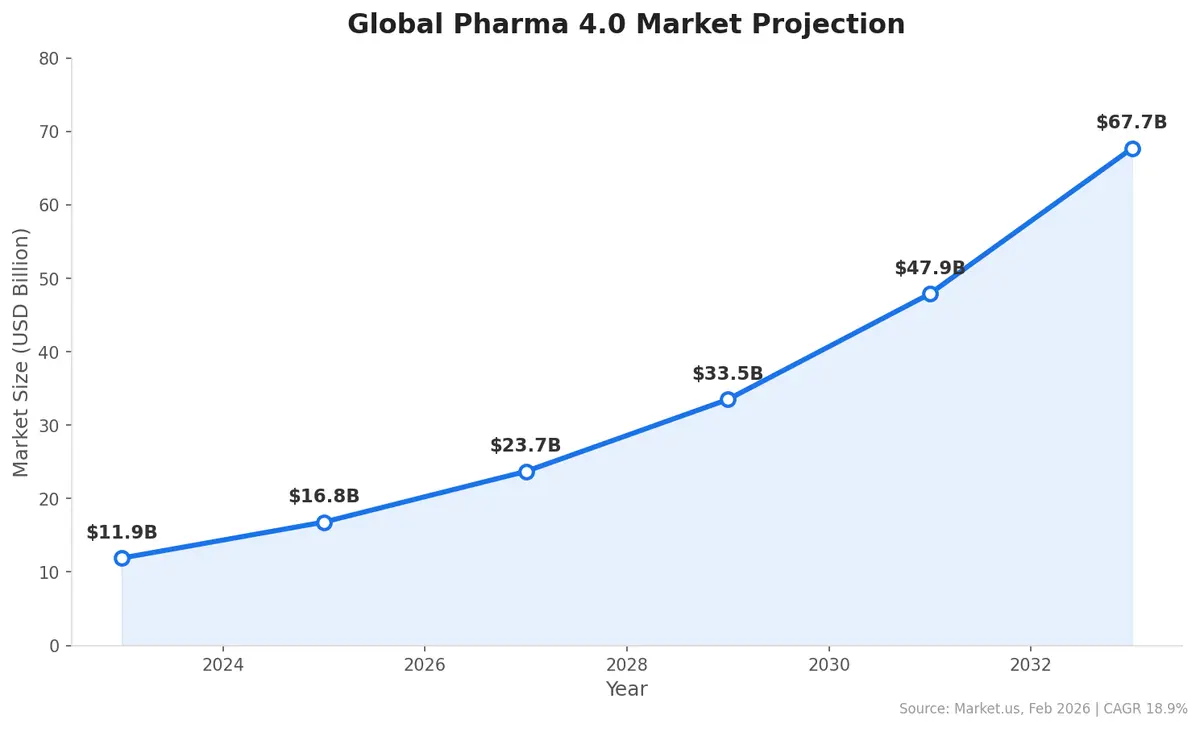
The broader context reinforces why this matters now. The global Pharma 4.0 market — encompassing AI, IoT, digital twins, and smart manufacturing in pharmaceuticals — is projected to grow from $11.9 billion in 2023 to $67.7 billion by 2033, a CAGR of 18.9%. AI in pharmaceuticals specifically is expected to reach $21.5 billion by 2035, growing at a 27% CAGR.
The pharmaceutical cold chain logistics segment alone was valued at over $11.2 billion in 2022 and is growing at 9.5% annually. As biologics, cell and gene therapies, and mRNA-based treatments continue to expand, the cold chain complexity that AI agents must navigate will only increase.
Meanwhile, counterfeit pharmaceuticals remain a persistent threat. Criminals increasingly target high-value biologics and weight-loss drugs, exploiting weak cold chain controls. Serialization combined with AI-powered anomaly detection creates a defensive layer that manual processes simply cannot match.
The Governed Agent Imperative
What makes pharma's approach to agentic AI instructive for the broader logistics industry is the emphasis on governance. These aren't autonomous agents making unchecked decisions. They operate within strictly defined regulatory guardrails, with every action auditable, every decision traceable, and every exception escalatable to human oversight.
TraceLink's OPUS platform delivered 127 standardized B2B integrations and four new enterprise system integrations in 2025, connecting with SAP, Oracle, Manhattan, and other major platforms. This integration ecosystem means AI agents don't operate in silos — they orchestrate across ERP, WMS, serialization, and pharmacy systems simultaneously.
The lesson for logistics operators outside pharma is clear: the most successful agentic AI deployments won't be the ones with the most powerful models. They'll be the ones with the most trusted data foundations and the most rigorous governance frameworks.
What This Means for Healthcare Logistics Operations
For shippers and logistics providers handling pharmaceutical products, the agentic orchestration wave creates both opportunities and imperatives:
-
Data readiness is prerequisite. Before deploying AI agents, ensure your transaction data is structured, standardized, and compliant. Garbage in, autonomous garbage out.
-
Multi-enterprise connectivity matters more than internal optimization. The value of agentic orchestration scales with the number of connected trading partners. Isolated AI deployments miss the point.
-
Governance is a feature, not a constraint. Regulated industries are proving that governed agents outperform ungoverned ones — because trust enables adoption at scale.
-
Cold chain and serialization are converging. Temperature monitoring, serialization verification, and logistics execution are increasingly managed as unified workflows rather than separate compliance silos.
The pharmaceutical industry's move toward agentic orchestration represents the most rigorous test case for AI in logistics. If governed agents can manage the complexity of serialized, temperature-sensitive, multi-enterprise pharmaceutical supply chains, the model will extend rapidly into other regulated verticals — food safety, chemicals, and high-value electronics among them.
Managing complex pharmaceutical or healthcare logistics? Contact CXTMS for a demo of how our TMS platform integrates compliance, cold chain visibility, and intelligent automation into unified supply chain workflows.


